Keywords:
Pancreas, Oncology, CT, Surgery, Fistula
Authors:
R. Valletta1, G. A. Zamboni1, M. C. Ambrosetti1, G. Marchegiani1, M. Bonatti2, G. Mansueto1; 1Verona/IT, 2Bolzano/IT
DOI:
10.26044/ecr2019/C-2050
Methods and materials
In this retrospective study we analysed the preoperative MDCTs of 48 patients who underwent pancreatico-duodenectomy for adenocarcinoma between January 2011 and June 2018.
24 patients (15 males,
9 women; ages 41-75,
mean age 62) developed a clinically relevant postoperative pancreatic fistula,
and 24 (15 males,
9 women; ages 34-76,
mean age 60) did not.
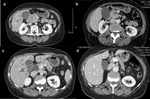
We included in this study only patients with histologically proven adenocarcinoma (Figure 1).
Exclusion criteria was the presence of significant motion artifacts.
All patients underwent a CT scan (Brilliance 64,
Philips),
before and after administration of high-concentration iodined contrast agent (1.5 ml / kg of 370 mg / ml contrast agent).
The contrast-enhanced phases were timed with a bolus-tracking technique,
by positioning a region of interest (ROI) in the abdominal aorta,
typically at the level of the celiac axis: the late arterial phase was acquired with a 15-s delay from the enhancement threshold (150 HU) and the portal-venous phase with a 60-s delay from the threshold.
One reader exported the portal-venous phase images in DICOM format from PACS to an independent workstation after patient-identifying informations were removed.
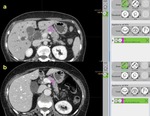
A minimum area of 10 mm2 of the pancreatic parenchyma at the resection margin (where the anastomosis would be performed),
was manually contoured encompassing as much of the pancreatic parenchyma as possible,
avoiding tumoral structures and the pancreatic duct in order to perform volumetric CT texture analysis (Figure 2).
Volumetric texture analysis was performed using a free software (LIFEx) [12].
For statistical analysis GraphPad Prism was used (GraphPad Prism version 7.00 for Windows,
GraphPad Software,
La Jolla California USA,
www.graphpad.com).
Results were compared between the two groups using Student’s t test.