Thymic function,
embryology,
histology and anatomy
The thymus is a central lymphoid organ that plays an important role in the maturation of the immune system,
especially regarding the cellular immunity during childhood [1].
The thymus arises in the sixth gestational week from the third and fourth branchial pouches and undergoes a descensus process towards its final location,
in the anterior mediastinum,
behind the sternum and anterior to the pericardium and great vessels,
and may extend longitudinally from the lower pole of the thyroid gland to the diaphragm [2].
At first,
the thymus gland is purely epithelial,
but in the tenth gestational week it is populated with lymphoid cells migrated from the fetal liver and bone marrow [3].
Histologically,
the cortex of the thymus consists mainly of lymphocytes,
whilst the medulla contains primarily epithelial cells. Other types of cells are also present,
such as myoid cells,
which may play a role in the pathogenesis of myasthenia gravis [3].
Thymic morphology has great variability.
In young adults,
it is frequently bilobated,
but may also be unilobated or trilobated [2].
Fig. 1
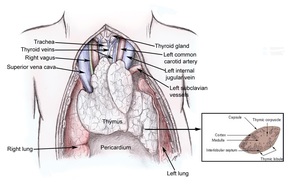
Fig. 1: Thymic morphology and histology [4, 5].
References: Said Fadi Yassin, (2018) Surgery of Thymus Gland. Medscape; Mark Hill (2018) SH Lecture- Lymphatic Structures and Organs. Embriology
As the function of the thymus decreases with age,
the epithelial component atrophies.
Throughout a process of fatty involution,
the shape of the gland modifies and its size and density decrease.
In the context of continuously changing thymic morphology,
it is important to be familiar with the CT aspects of the normal thymus and its main pathology.
CT protocol in the evaluation of the thymus gland
In the evaluation of the mediastinum,
CT is the standard imaging technique. It is commonly performed as part of a general thoracic examination. The protocol requires thin-section multislice acquisition (0.75- 1.5 mm) with reconstructed axial images of 5 mm and multiplanar reformation.
Noncontrast and contrast-enhanced CT is indicated to evaluate a mediastinal mass,
in purpose to appreciate enhancement characteristics of the lesion and vascular invasion [6,7].
For adults,
intravenous non-ionic iodinated contrast media should be injected in volumes of 100-150 ml,
at flow rates of 3-5 ml /sec in order to evaluate tumor vascularity and mediastinal vessels [6].
Thymic pathology
Anterior mediastinal pathology includes a wide variety of lesions.
The most common entities can be summarized using the mnemonic “4T” which stands for:
- thymoma,
- teratoma,
- thyroid tumors/ goiter and
- terrible lymphoma
Anterior mediastinal pathology involving the thymus includes also:
- thymic hyperplasia,
- cysts,
epithelial tumors,
- germ cell tumors and
- hematologic malignancies.
1.
Thymic hyperplasia
There are two histological forms of thymic hyperplasia: true hyperplasia and lymphoid hyperplasia.
True thymic hyperplasia,
or rebound hyperplasia,
is frequently related to chemotherapy,
radiation or corticosteroid therapy,
major surgery,
burns or infections [1].
Under stress,
the thymus becomes atrophic.
After the cessation of the infliction,
frequently in 6-9 months,
it grows back to the original size,
or even larger [7].
Lymphoid hyperplasia is characterized by an increased number of lymphoid follicles in the medulla and is associated with immunologically mediated disorders such as myasthenia gravis (MG),
systemic lupus erythematosus,
rheumatoid arthritis or Graves’ disease [1].
2.
Thymic cyst
Thymic cysts may be congenital or acquired through thoracotomy or cystic changes in thymic epithelial tumors,
lymphomas or germinal cell tumors [2].
3.
Thymic epithelial tumors
Thymic epithelial tumors arise from thymic epithelium and include noninvasive/invasive thymoma and thymic carcinoma.
Whilst noninvasive thymoma is an encapsulated tumor without capsular effraction,
invasive thymoma and thymic carcinoma may exhibit local invasiveness,
lymphadenopathies and metastases.
The WHO classification of thymic epithelial tumors is a histologic classification and correlates with prognosis [10].
| WHO Classification |
Suster- Moran Classification |
| Type- A thymoma |
Thymoma
|
| Type- AB thymoma |
| Type- B1 thymoma |
| Type- B2 thymoma |
| Type- B3 thymoma |
Atypical thymoma |
| Type- C thymoma |
Thymic carcinoma |
Table 1.
WHO classification of thymic epithelial tumors [10].
The Masaoka staging system is based on the presence of invasion and also correlates with the prognosis [7].
| Stage |
Characteristics |
| Stage I |
Micro- and macroscopically encapsulated. |
| Stage IIa |
Macroscopic invasion of the surrounding fatty tissue or
mediastinal pleura.
|
| Stage IIb |
Microscopic invasion of the capsule. |
| Stage III |
Macroscopic invasion of the pericardium,
great vessels or lung. |
| Stage IVa |
Pleural or pericardial dissemination. |
| Stage IVb |
Lymphogenous or hematogenous metastatic disease. |
Table 2. Masaoka staging system [7].
4.
Germinal cell tumors
Germinal cell tumors originate from multipotent primitive germ cells.
These tumors mainly arise in the gonads,
but may also be present in the midline of the body,
from the pineal gland to the sacrococcygeal region, arising from germ cells misplaced during their migration from the yolk endoderm to the gonads in embryogenesis [11].
The most common extragonadal site is the anterior mediastinum,
and the average age of presentation is between the second and fourth decade of life [7].
Germ cell tumors can be subdivided into benign lesions,
such as mature teratoma (the most frequent subtype) and malignant tumors; seminoma and nonseminomatous germ cells tumors [7].
Malignant tumors occur mostly in men and may secrete tumor markers: human chorionic gonadotropin (hCG) in pure seminomas and α-fetoprotein (AFP) in nonseminomatous germ cell tumors [11].
5.
Thymic lymphoma and leukemia
Thymic involvement usually occurs secondary to widespread disease,
but may also be isolated.
The thymus gland is more commonly affected in Hodgkin disease,
frequently in young patients (average age of 30 years) [2].
| Pathology |
CT aspects |
Diagnostic features |
|
Thymic
hyperplasia
|
Symmetrical enlargement of the thymus,
NO contour irregularities or focal masses. |
Differentiation between thymic hyperplasia and neoplasms on CT alone is not possible in some cases; chemical shift MRI might be useful. |
| Thymic cyst |
Fluid/parafluid (may exhibit higher densities),
well outlined mass ± wall calcifications. |
MRI should be performed [9]. |
| Thymoma |
Round/ lobulated,
well outlined,
homogenous/ slightly heterogeneous mass,
± calcifications. |
Clinical aspects: MG or other paraneoplastic syndromes.
Biopsy may be required [9].
|
|
Invasive
thymoma
|
Lobulated,
homogeneous/ slightly heterogeneous mass,
local invasiveness. |
Requires biopsy [9]. |
|
Thymic
carcinoma
|
Large,
heterogeneous mass,
locally invasive,
lymphadenopathies or distant metastases. |
Requires biopsy [9]. |
| Teratoma |
Heterogeneous mass with fat,
fluid,
soft tissue and calcar components. |
Imaging alone may suffice [9]. |
| Seminoma |
Large,
lobulated,
homogenous mass with local invasiveness,
lymph node and systemic metastases. |
Clinical context: young male.
Laboratory findings: hCG.
Biopsy is required [9]. |
|
Nonseminomatous
germ cell tumor
|
Large,
lobulated,
heterogeneous mass with local invasiveness and lymphogenous and hematogenous metastases
|
Clinical context: young male.
Laboratory findings: AFP.
Biopsy is required [9]. |
| Lymphoma |
Lobulated mass with mild contrast enhancement,
local invasiveness,
surrounding the great vessels but WITHOUT invasion.
Lymphadenopathies. |
Clinical: “B” symptoms.
Cytology and biopsy [9]. |
Table 4: Differential diagnosis amongst thymic masses.

![Fig. 1: Thymic morphology and histology [4, 5]. References: Said Fadi Yassin, (2018) Surgery of Thymus Gland. Medscape; Mark Hill (2018) SH Lecture- Lymphatic Structures and Organs. Embriology](https://epos.myesr.org/posterimage/esr/ecr2019/147730/media/794405?maxheight=150&maxwidth=150)

