Learning objectives
To review non-HCC malignancies arising in the cirrhotic liver
To identify the imaging criteria of LI-RADS M on all modalities
Review literature on LR-M performance and pathological entities
Background
The most common liver cancer in cirrhosis is Hepatocellular Carcinoma (HCC), patients with cirrhosis are also at increased risk for developing intrahepatic cholangiocarcinoma (iCCA) and combined HCC-CCA. Some other malignant lesions may also exist or coexist with HCC.While non-HCC malignancies often occur in patients without cirrhosis, their incidence is increasing in cirrhotic livers.
The American College of Radiology recommends theLiver Imaging Reporting and Data System (LI-RADS)to standardize the reporting and data collection of CEUS, CT-MRI for HCC. LI-RADS-M has specific criteria that allow for the...
Findings and procedure details
CEUS LR-M Criteria
Example:Fig. 5
CEUS Ancillary Imaging Features
CT-MR LR-M Criteria
Examples:Fig. 8,Fig. 9, Fig. 10,Fig. 11
CT-MRI Ancillary Imaging Features
The next algorithm suggests a probable etiology based on radiological findings.
The differential diagnosis includes; atypical HCC, iCCA, cHCC-CCA, and less common, primary tumor, metastases, and benign entities.
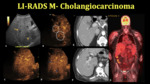
Intrahepatic CCA
CCA is the most common biliary tract malignancy and the second most common primary hepatic tumor after HCC.
Among numerous risk factors described for iCCA, many are similar to those reported for HCC....
Conclusion
There are lesions thatare being seen with increasingincidence in cirrhotic patients such as iCCA, cHCC-CCA, and atypical HCC. Primary liver lesions are rare. Metastatic disease is also rare in the setting of cirrhosis.
Often biopsy is indicated to diagnose LR-M lesions.
The radiologist needs to know that not all lesions in the cirrhotic liver are HCC, its necessary to see the dark side of the liver and look for the atypical malignancies in cirrhosis.
Personal information and conflict of interest
P. M. Dautt
[email protected]
B. Sirlin.Disclosures.Industry research support:Bayer,GE,Gilead,Philips, Siemens.Institutional Consulting Representative:BMS,Exact Sciences,IBM-Watson.Consulting:Blade,Boehringer,Epigenomics.Lab service agreements:Enanta,Gilead,ICON,Intercept,Nusirt,Shire,Synageva,Takeda
K. Fowler.Disclosures.Industry research support:Bayer,GE, Philips,Siemens.Consulting:Bayer, Epigenomics,Medscape,12 Sigma
References
ACR(2018)CT/MRI LI-RADSv2018core-ACR.(2017)CEUS LI-RADSv2017core.
Kamath, A.,Roudenko, A.,Hecht, E.,Sirlin,C.,Chernyak,V.,Fowler,K.,& Mitchell, D. G. (2019).CT/MR LI-RADS2018:clinical implications and management recommendations.Abdominal Radiology,44(4),1306-1322.
Chung, Y. E., Kim, M. J., Park, Y. N., Choi, J. Y., Pyo, J. Y., Kim, Y. C., ... & Choi, S. Y. (2009). Varying appearances of cholangiocarcinoma: radiologic-pathologic correlation.Radiographics,29(3), 683-700.
Fábrega-Foster,K.,Ghasabeh, M.A., Pawlik, T. M., & Kamel, I. R. (2017). Multimodality imaging of intrahepatic cholangiocarcinoma.Hepatobiliary surgery and nutrition,6(2), 67.
Stavraka,C.,Rush,H., & Ross, P.(2019).Combined hepatocellular cholangiocarcinoma (cHCC-CC):An update of genetics, molecular biology, and therapeutic interventions.Journal of hepatocellular carcinoma,6,11....