Purpose
Carfilzomib is a novel, irreversible inhibitor of the proteasome’s chymotrypsin-like activity (see Figure 1). It is currently broadly used in relapsed or refractory multiple myeloma patients (1,2). Early, preclinical and clinical data point towards a cardiotoxic side effect due to the fast and highly efficient inhibition of the proteasome in cardiomyocytes (2). Although this drug is known to be cardiotoxic, so far there are only limited data about assessment of myocardial damage using cardiac MRI (CMR) and early identification of patients at risk.
Therefore, this...
Methods and materials
Patients (see Table 1) diagnosed with multiple myeloma and scheduled to undergo treatment with Carfilzomib have been recruited and performed three CMR examinations using clinical 1.5T and 3T MRI systems (Philips Ingenia_CX and Ingenia, Best, Netherlands). Scans were performed before onset of therapy (baseline) as well as 3-5 days and 3 months thereafter.
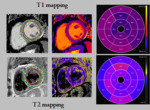
The CMR parameters included standard morphological and functional parameters for left and right ventricles (LV/RV) i.e. ejection fraction (EF), enddiastolic volume/body surface area (EDV/BSA), endsystolic volume/BSA (ESV/BSA), mitral and tricuspid annular plane...
Results
So far, we have enrolled 14 patients. Two patients dropped out from the 2ndfollow up due to change of chemotherapy regimen or death.
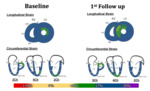
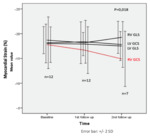
We found that the mean global LV Strain parameters were within normal range at baseline and stable during follow-up. Five patients however showed an increase in the number of abnormal LV segments between baseline and follow-up scans(seeFigure 3).Furthermore, at the 2ndfollow up three of those patients showed a decrease of LVEF and/or MAPSE which was associated with the appearance of clinical symptoms...
Conclusion
Our preliminary results indicate that Carfilzomib may affect ventricular strain in the absence of detectable tissue pathology. Strain parameters may provide sensitive markers to identify an impact of chemotherapy on the myocardium at an early stage and thus develop preventive strategies.
Personal information and conflict of interest
N. A. Atia, M.B.B.Ch., MSc.
Assistant Lecturer of Diagnostic Radiology, Tanta University Hospitals, Egypt.
Research Fellow at Cardiology Department, Heidelberg University Hospital, Germany.
Phone: +4917636539121
e-mail :
[email protected]
References
Siegel D. S., Martin T., Wang, M., et al: A phase 2 study of single-agent carfilzomib (PX-171-003-A1) in patients with relapsed and refractory multiple myeloma. BLOOD Journal. 2012. 120 (14): 2817-2825. DOI:10.1182/blood-2012-05-425934.
Kim G. Y., Ahuja T., Papadopoulos J. and Cirrone F.: Cardiotoxicity with carfilzomib at doses greater than 27 mg/m2: A case series. Journal of Oncology Pharmacy Practice. 2017. 0(0): 1–5. DOI:10.1177/1078155217729564.
Kortuem, M.K. and Stewart, K.A.: Carfilzomib. The American Society of Hematology. 2013. DOI 10.1182/blood-2012-10-459883.