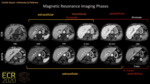
Purpose
The adequacy of Portal Venous Phase (PVP) on contrast-enhanced CT and MR
imaging studies, is crucial for the evaluation of hepatic enhancement and detection of focal liver lesions [1]. According to EASL guidelines and LI-RADS, washout detected on PVP or delayed phase represents one of the major features for the characterization of hepatocellular carcinoma (HCC) [2].
The use of hepatocyte-specific contrast agents, such as gadoxetic acid and gadobenate dimeglumine, has improved the diagnostic accuracy of MR for the diagnosis of HCC [3]. Some studies have...
Methods and materials
The local institutional review board approved this retrospective cross-sectional study.
We included cirrhotic patients who underwent both gadoxetic acid and gadobenate dimeglumine enhanced abdominal MRI exams. Contrast-enhanced abdominal MRI exams were performed with two different 1.5-T MR systems using appropriate liver protocols. We ruled out exams with MRI artifacts and average time between the two exams greater than 24 months. For each exam, two readers independently measured signal intensities of liver parenchyma (4 ROIs), and psoas muscle (1 ROI) on pre- contrast and portal venous...
Results
Study population included 84 cirrhotic patients (mean age ±SD: 66 ± 13 years old) (Figure 5).
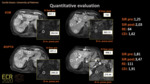
The relative enhancement of cirrhotic liver parenchyma was significantly higher with gadobenate dimeglumine (Reader 1: 56.08 ± 58.84; Reader 2: 57.74) compared to gadoxetic acid (Reader 1: 42.38 ± 44.58; Reader 2: 42.39 ± 42.90) (Reader 1: p=0.044; Reader 2: p=0.027), with almost perfect inter-reader agreement (ICC:0.94)(Figure 6).Signal intensity and signal intensity ratio of liver parenchyma as well as contrast enhancement index were not significantly different (Figure 7).
Due...
Conclusion
In cirrhotic patients, there are significant changes in the enhancement of liver parenchyma on portal venous phase using the two commercially available hepatobiliary contrast agents, with higher relative enhancement of liver parenchyma with gadobenate dimeglumine compared to gadoxetic acid.
Personal information and conflict of interest
C. Gozzo; Palermo/IT - nothing to disclose.
[email protected]
F. Vernuccio; Palermo/IT - nothing to disclose.
V. Greco; Palermo/IT - nothing to disclose.
R. Cannella; Palermo/IT - nothing to disclose.
M. Midiri; Palermo/IT - nothing to disclose.
G. Brancatelli;Palermo/IT - nothing to disclose.
References
European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol 2018; 69:182-236.
Kambadakone AR, Fung A, Gupta RT, et al. LI-RADS technical requirements for CT, MRI, and contrast-enhanced ultrasound. Abdom Radiol (NY) 2018; 43:56-74.
Vernuccio F, Cannella R, Meyer M, et al. LI-RADS: Diagnostic Performance of Hepatobiliary Phase Hypointensity and Major Imaging Features of LR-3 and LR-4 Lesions Measuring 10-19 mm With Arterial Phase Hyperenhancement. Am J Roentgenol 2019; 213(2):W57-W65.
Frydrychowicz A, Nagle SK, D'Souza SL, Vigen...