Renal transplant recipients undergo close imaging surveillance. Ultrasound (particularly Doppler ultrasound) is the first-line imaging modality chosen to evaluate these patients, especially in the early postoperative period. However, CT is also useful in diagnosing complications.
Renal transplant complications can be categorized as vascular and non-vascular. This exhibit will focus on the latter (Fig. 2).

Fig. 2: Non-vascular complications of renal transplantation.
Non-vascular complications
Renal parenchymal abnormalities
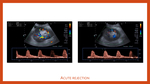
Acute tubular necrosis (ATN) generally occurs in the immediate posttransplant period and is related to ischemia of the allograft before vascular anastomosis (Fig. 3). Most episodes of ATN resolve spontaneously and appear to not impact ultimate graft survival.
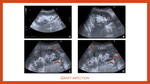
Graft rejection continues to represent a significant source of morbidity. Hyperacute rejection is very rare and usually manifests during surgery. These grafts are rarely salvageable. Accelerated acute rejection occurs in the first week of transplantation. Acute rejection mostly occurs during the first 10 weeks after surgery. Ultrasound may show an oedematous kidney with thickened cortex, loss of corticomedullary differentiation and thickening of the collecting system walls (Fig. 4). Chronic rejection may appear months to years after transplantation and its generally irreversible. Ultrasound usually shows a small kidney with thinning and increased echogenicity of the cortex (Fig. 5, A and B).
Nephrotoxicity secondary to drugs (particularly calcineurin inhibitors) has sonographic findings similar to rejection.
On colour Doppler ultrasound and spectral analysis, patients with these abnormalities may reveal diminished cortical flow and increased intraparenchymal arterial resistive indices (RI) (Fig. 5C and Fig. 6), but RI greater than 0,80 may be seen in other settings including ureteral obstruction, renal vein thrombosis, and mass effect on the allograft.
These parenchymal abnormalities are generally not distinguishable based on ultrasound, and the role of imaging is to rule out other causes of graft failure. A biopsy may be performed to establish a definitive diagnosis.
Graft infection is common within the first 4 months after transplantation, and some patients may develop frank sepsis. Symptoms may not be as obvious in transplanted patients, but the imaging findings of pyelonephritis in renal allografts are similar to those encountered in native kidneys. Ultrasound is often normal but may show enlargement of the kidney, altered renal parenchymal echogenicity with focal wedge-shaped areas of hyper or hypoechogenicity (Fig. 7) and urothelial thickening. Contrast-enhanced CT may demonstrate wedge-shaped areas of hypoperfusion (Fig. 8) or a striated nephrogram. Perinephric stranding may also be found. Most importantly, the role of imaging is to rule out complications such as abscess formation.
Urological abnormalities
Urinary leak generally occurs in the first 3 months after transplantation and it originates most commonly from the ureterovesical anastomosis, followed by the vesicostomy site and the transplanted renal pelvis. If the leak is small and temporary, the urine is usually resorbed; if it endures, a urinoma usually forms. CT urography may diagnose an acute urine leak by showing extravasation of intravenous contrast material during the excretory phase (Fig. 9 and Fig. 10).
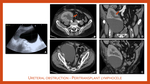
Ureteral obstruction has a bimodal distribution. Less frequently, it can occur in the first few days after surgery due to ureteral kinking, ischemia, or external compression from postoperative oedema or hematoma. Ureteral obstruction is far more common after the first month. It is usually caused by ureteral stricture due to ischemia or scar tissue, extrinsic compression from a collection (Fig. 11), or less often intraluminal abnormalities such as a stone. Ultrasound shows a dilated renal collecting system (Fig. 11, A and B). Ureteral dilatation to the bladder may be seen when there is an anastomotic stricture (Fig. 12). Evaluation at ultrasound should include a thorough assessment of the collecting system and transplant ureter, noting the presence of fluid collections, intraluminal debris or stones. CT can be helpful in identifying the cause and level of obstruction as the ureters can be difficult to follow on ultrasound.
Perinephric fluid collections
Perinephric fluid collections may represent hematomas, urinomas, abscesses or lymphoceles. Imaging features at grey-scale ultrasound may be similar, but differentiation can be achieved by considering the time from surgery, clinical manifestations and the use of colour Doppler ultrasound.
Perinephric hematomas are common in the immediate postoperative period. Ultrasound shows a complex heterogeneous perinephric collection (Fig. 13 and Fig. 14). Nonenhanced CT demonstrates a fluid collection with attenuation values greater than 30 Hounsfield units (Fig. 14).
Urinomas also develop in the early postoperative period, from acute urinary leaks. Ultrasound and CT typically demonstrate a fluid collection between the transplanted kidney and the bladder. The fluid in urinomas appears echo-free on ultrasound (Fig. 15) and has a density near water on CT, which may be difficult to distinguish from a seroma or lymphocele. Delayed contrast-enhanced CT to detect leakage of excreted contrast material allows differentiation.
Perinephric abscesses are uncommon complications that manifest during the first weeks to months after transplantation. They usually arise as a complication of pyelonephritis, although they may occur as a direct complication of surgery or as a secondary infection of a previous fluid collection. CT and ultrasound are the most useful imaging techniques, but abscesses are often indistinguishable from sterile collections on imaging. Fever and leucocytosis should raise suspicion. Perirenal abscesses can appear as irregular thick-walled fluid collections with peripheral hyperaemia and surrounding inflammatory change in the peritransplant fat. Ultrasound typically shows a complex fluid collection which may be nearly anechoic with few internal echoes or hypoechoic with heterogeneous internal echoes (Fig. 16). Sometimes, they are multiloculated and may have blood flow present in the thickened wall and septa at colour Doppler assessment. On unenhanced CT scans, abscesses are hypoattenuating fluid collections which may contain areas of hyperattenuation (Fig. 17, A). On contrast-enhanced scans, a peripheral rim-like enhancement may be present (Fig. 17, B). Occasionally, foci of gas within the fluid collection will be found.
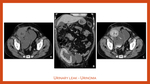
Lymphoceles are the most common peritransplant fluid collections (1% to 15% of transplant recipients). They most frequently occur 2 weeks to 6 months after surgery. Ultrasound typically shows well marginated anechoic or hypoechoic fluid collections occasionally containing thin internal septa (Fig. 11,A; Fig. 18). They may be indistinguishable from urinomas or seromas on ultrasound. CT demonstrates well-defined hypoattenuating fluid collections with sharp borders that usually have an attenuation value near water (Fig. 11, B-E).
Post-transplant malignancies
Post-transplant malignancies are long-term complications for which recipients are at increased risk due to prolonged immunosuppression. The risk of malignancy is 3 to 5 times higher in renal transplant recipients compared to the general population, particularly nonmelanomatous skin cancer, lymphoma, and colon cancer. The risk of primary renal malignancy in the allograft is about six times that in the native kidney.
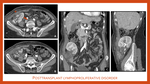
Posttransplant lymphoproliferative disorder (PTLD) occurs in 1 to 3% of renal allograft recipients and is associated with Epstein-Barr viral infection. When it occurs in renal transplant patients, the allograft is the most common site of involvement (Fig. 19), and as a tendency to manifest as a mass replacing or encasing the hilum, leading to outflow obstruction. Lymphadenopathy is the most common finding on CT (Fig. 20). In more advanced stages of the disease, solitary or multiple low-attenuation masses involving the allograft and other solid organs can be seen.