MRI features of GP
GP lesions tend to originate from the peripheral zone and may be diffuse, nodular or occasionally multifocal(6).
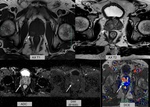
•On T2 weighted images(T2WI); GP lesions appear hypointense and occasionally can cause capsular irregularity and/or capsular bulging(Fig. 1) which are suggestive of extracapsular extension (1,7).

Fig. 1: Right PZ lesion(arrows), diagnosed with GP pathologically. Note that this lesion causes bulging and irregularity of the overlying capsular margin on T2WI(arrowhead). Also, note the hyperintensity on T1WI(dashed arrow).
References: Diagnostic Radiology, American Hospital - Istanbul/TR
•On diffusion-weighted images (DWI); GP lesions may show hyperintensity on high b-value diffusion images and low apparent diffusion coefficient (ADC) values consistent with impeded diffusion, similar to that of prostate cancer (1,7). It is supposed that impeded diffusion findings on DWI and ADC maps are associated with the degree of acute inflammation and the existence of caseating necrosis(6,8,9).

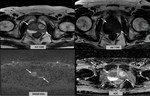
Fig. 3: Left PZ and right transition zone(TZ) lesions(arrows) which have similar appearance with prostate cancer on mp-MRI. These lesions diagnosed with GP pathologically. Note the hyperintensity on T1WI(dashed arrow).
References: Diagnostic Radiology, American Hospital - Istanbul/TR

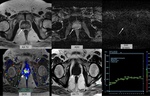
Fig. 4: Pathologically proven GP lesion at right PZ(arrows). This lesion appears hypointense on T2WI and shows slight-moderate diffusion restriction on high b-value diffusion series. Note the prolonged enhancement on DCE(arrowhead). Also, note the hyperintensity on T1WI(dashed arrow).
References: Diagnostic Radiology, American Hospital - Istanbul/TR
•On dynamic contrast-enhanced(DCE); series, GP lesions show different enhancement patterns depend on histopathological features. GP lesions with caseation necrosis or abscess formation show early and prolonged ring enhancement while non-caseating GP lesions tend to show early enhancement and washout kinetics (1,4).

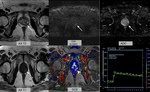
Fig. 2: Left PZ lesion(arrows) which shows hypointensity on T2WI and marked diffusion restriction on DWI, consistent with prostate cancer. Note the rapid enhancement on DCE images(arrowhead). Also, note the hyperintensity on T1WI(dashed arrow). Pathology revealed non-caseating GP.
References: Diagnostic Radiology, American Hospital - Istanbul/TR

Fig. 4: Pathologically proven GP lesion at right PZ(arrows). This lesion appears hypointense on T2WI and shows slight-moderate diffusion restriction on high b-value diffusion series. Note the prolonged enhancement on DCE(arrowhead). Also, note the hyperintensity on T1WI(dashed arrow).
References: Diagnostic Radiology, American Hospital - Istanbul/TR
These imaging features on mp-MRI cause a diagnostic dilemma between prostate cancer and GP. Therefore, there are some limited studies based on small patient groups diagnosed with GP. These studies have identified some features on mp-MRI that may differentiate GP from prostate cancer.
-
Naik et al(10); were among the first to look at MRI features of GP who investigated the transrectal ultrasound and MRI(T2WI and T1W-precontrast and postcontrast images) features of BCG-induced GP, reported that there isn’t any clinical, laboratory or radiological feature which help differentiation GP from prostate cancer.
-
Bour et al(11); defined two types of GP MRI appearances; first type, the frequently reported tumor-like appearance that cannot be differentiated from prostate cancer and the second type, a rarer appearance consistent with caseous necrosis and abscess formation.
-
Suzuki et al(9); performed biparametric MRI in 10 patients and they described three main patterns of GP which were diffuse(Fig. 5), nodular ( Fig. 2 ) and cystic with mural nodule. The diffuse pattern was the most common. Furthermore, they stated that diffuse lesions were hyperintense on T1WI.
Our mp-MRI findings
We retrospectively reviewed 5 patients with pathologically proven GP and included in this study. Patients we referred for either rising PSA or abnormal DRE. The median age was 74.8(range 72-76) years. Median PSA was 6.35 ng/ml (range 4.11-9.79 ng/ml). All patients had a history of BCG treatment.
Our mp-MRI findings(table) were consistent with previous studies emphasizing the radiological overlap between GP and prostate cancer.
On mp-MRI; all patients were assigned a Prostate Imaging Reporting and Data System(PI-RADS) 4 or 5 suspicion score and one patient(Fig. 1) had suspicious findings for extracapsular extension. MRI guided in-bore biopsy was performed in all of these patients due to high suspicion for prostate adenocarcinoma.
A summary of GP mp-MRI findings of our patients shown in the table.
|
Case
|
Age
(years)
|
BCG
history
|
Location
|
Shape
|
PSA
(ng/ml)
|
SI on T1WI
|
SI on T2WI
|
DWI
|
DCE
|
|
1
|
76 |
+ |
PZ
|
nodular |
5.2 |
high |
low |
high |
markedly |
|
2
|
73 |
+ |
PZ |
nodular |
4.11 |
high |
low |
high |
markedly |
|
3
|
74 |
+ |
PZ |
nodular |
6.45 |
high
(slightly)
|
low |
high
(slightly)
|
prolonged |
|
4
|
76 |
+ |
PZ & TZ |
nodular |
9.79 |
high
|
low |
high
|
markedly |
|
5
|
72 |
+ |
PZ |
diffuse |
6 |
high
(slightly)
|
low |
high |
markedly |

Fig. 7: T1W ımages of all lesions which have similar appearance with prostate cancer on mp-MRI and diagnosed with GP pathologically. Note the hyperintensity(arrows) of all lesions.
References: Diagnostic Radiology, American Hospital and Koc University Hospital - Istanbul/TR