The present work employs a script from the REMbox framework (Dicom Port AB,
Umeå,
Sweden) written in Python™ where the patient contour is automatically segmented,
i.e.
removing the patient table and mattress.
Based on recommendations from the Report of AAPM Task Group 220[2],
the script then use the segmented patient volume to determine the WED.
To validate script functionality with regard to segmentation,
determination of WED and centring errors,
a Catphan® 500 was used (The Phantom Laboratory,
USA).
Phantom images were evaluated from known size and material constitution of Catphan® 500,
as well as centring from placing the phantom at isocentre and ±30 mm off centre using the patient table.
All verification results were within one pixel,
i.e.
errors less than 1.2 mm.
The image material used in this work consisted of 218 thorax examinations in the inspiration phase performed with a Siemens Definition Flash (Siemens Healthineers,
Germany),
see Table 1 for examination protocol details.
All examinations were performed with a single CT scanner at the University Hospital of Umeå,
Sweden.
Table 1. Thorax examination protocol setting for the Siemens Definition Flash used in this work.
| Data collection (mm) |
128 x 0.6 (38.4 mm nominal) |
| Pitch |
0.9 |
| Tube voltage (kVp) |
120 |
| Rotation time (s) |
0.5 |
| Reconstructed slice thickness (mm) |
5 |
| Iterative reconstruction |
SAFIRE 4 |
| Reconstruction filter |
I31f medium smooth |
| TCM,
CARE Dose 4D |
Quality Ref mA 25 |
| Field of view,
FOV (cm) |
50 |
A single slice was chosen from a specific reconstruction over the liver and sent from our PACS solution (Sectra,
Sweden) for automatic evaluation.
These CT images were chosen as a test of the script because they are heterogeneous,
containing air from the lungs,
a lot of soft tissue,
as well as bone from the spine and the rib cage.
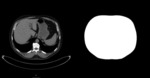
The segmentation script automatically yields an image mask,
delineating the segmented volume.
These masks were manually evaluated for each examination to determine if segmentation had been successful.
An example of original image and segmentation mask result is shown in Fig.
2.
When the patient contour has been segmented,
the WED can be determined using the Hounsfield units in the reconstructed image slice.[2] From the WED,
the Python™ script used in this work automatically determines the appropriate correction factor,
f,
from the Report of AAPM Task Group 204.[1] Shown in Fig.
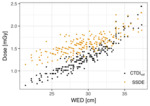
3 below are calculation results for SSDE as a function of WED in comparison to examination reported CTDIvol.
As seen in Fig.
3,
for smaller patient sizes the calculated SSDE are consistently greater than CTDIvol per patient WED.
As patient size increases,
the SSDE are eventually equal to or greater than the CTDIvol.
This is as expected according to the functionality of the SSDE,
as discussed in the Report of AAPM Task Group 204.
For body examinations,
the SSDE should increase dose (f > 1) for a patient size equivalent to less than a 32 cm diameter cylinder of PMMA,
i.e.
the standard body CTDI phantom.
Supawitoo et al.
reported an average Hounsfield value for a 32 cm diameter CTDI phantom as approximately 135 HU.[3] This corresponds to a WED of 34 cm.
As seen in Fig.
3,
this is approximately where the SSDE and CTDIvol results in the present work converge.
There is substantial scatter in the results for SSDE shown in Fig.
3,
more so than for the CTDIvol.
Two major contributions to this scatter can logically be identified.
First we have the heterogeneity of patient constitution in the image volume investigated.
By experiment design,
these images were chosen to make WED calculations on a volume containing different tissue types,
from sparse (lung tissue and air-filled intestine) to approximately water equivalent (liver and adipose tissue) and dense (bone).
However,
there is also variability between patients,
i.e.
amount of different tissue types,
as observed in the images while preparing this work.
Furthermore,
patient elliptical eccentricity,
ε,
which is defined as
√1-(AP/2)²/(LAT/2)²,
where LAT and AP are the patient lateral and anterior-posterior dimensions,
respectively,
will also contribute to the spread in SSDE results.
Shown in Fig.
4 are the SSDE results as a function of WED grouped by eccentricity.
As seen in Fig.
4,
there is a clear grouping of SSDE results by patient eccentricity.
A high eccentricity,
meaning more pronounced elliptical shape (red dots),
is clearly distinguishable from a medium-low eccentricity (green),
meaning more circular shape.
The intrinsic variability in tissue composition between patients used in this study has not been removed from the data in Figure 4,
i.e.
patient eccentricity is a major contributor to the scatter in SSDE results.
As previously discussed,
the WED should be superior to a simple metric of patient physical size.
Commonly an equal area diameter (EAD) is used to describe patient physical size,
i.e.
describing the elliptical shape of a patient as an equal area circle.
This is also done for the WED in the present work,
i.e.
normalizing results to a circular shape.
Shown in Fig.
5 is a comparison of WED and EAD for the thorax examinations investigated.
As shown in Fig.
5,
the EAD for the examinations investigated is consistently greater than the WED.
This means that the EAD overestimates patient size from the images used,
as compared to attenuation properties given by the WED.
Consequently,
SSDE based on the EAD would underestimate patient dose compared to a WED based approach.
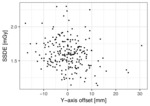
The Python™ script used to determine SSDE also calculates patient centring offset from the CT gantry isocentre.
Shown in Fig.
6 is the relation between SSDE and patient centring in the vertical direction (Y-axis offset).
As shown in Fig.
6,
there is a lot of scatter in the data describing relationship between SSDE and Y-axis offset.
The patients examined were predominantly well centred,
with a few outliers up to 30 mm offset.
Using statistical analysis no correlation between patient dose and Y-axis offset could be found.
This is not a proof of that patient centring within 30 mm does not yield additional patient dose.
The possible influence of patient centring on patient dose cannot be distinguished from the variance in SSDE resulting from patient constitution variability and eccentricity,
as previously discussed.