In our institution from 2007 to December 2018 the MDCT was performed annually in up to 400 cases of neonates with CHD.
CT scans were taken in all patients using a Somatom Sensation Cardiac 16 until 2016,
Somatom definition AS+ in 2016 and 2017,
and then Somatom Definition Edge in 2018 (Siemens AG,
Forchheim,
Germany).
The thorax is a low-attenuation region,
although substantial dose reduction during chest CT is feasible because of the high inherent contrast.
In our centre,
we decided to apply the ALARA principle as far as possible in neonates and babies with CHD
CT protocols:
- For evaluation of CHD,
electrocardiographic gating is not needed,
we never use it.
- Limit scan range to region of interest
Only one phase acquisition
- We need only arterial phase
- Unnecessary multiphase exams add substantial radiation dose
- We don’t need non-contrast scans
CT parameters
- Increase of collimation (you don’t need very thin slices) if you have 16 slices CT scan
- Lower kVp,
mAs
• Without electrocardiographic gating or respiratory arrest.
• Use iterative reconstruction
Technical parameters used for MDCT examinations in our institution were as follows for 16 slices CT:
- detector collimation,
1.5 mm;
- slice thickness,
2 mm;
- Increment,
1.5 mm;
- feed / rotation,
18 mm/sec;
- gantry rotation time,
0.5 sec;
- we always use 80 kVp as the standard kilovoltage setting in neonates and children up to 75 kg body weight;
- and current exposure is 40 mAs for all neonates.
Average CTDI vol was within 11 mGy,
DLP - 20 mGy x cm,
the average effective dose equivalent - 0.7 mSv
For the 128 slices CT technical parameters were as follows:
- detector collimation 128x 0,6 mm;
- slice thickness,
1 mm;
- Increment,
0,8 mm;
- feed / rotation,
230 mm/sec;
- gantry rotation time,
0.28 sec;
- we always use 70 kVp as the standard kilovoltage setting in neonates;
- In all scans,
dose modulation (CARE Dose 4D) was used to minimize the radiation dose.
- and current exposure is up to 20mAs for all neonates.
Average CTDI vol was within 3 mGy,
DLP - 5 mGy x cm,
the average effective dose equivalent - 0.2mSv.
Injection Protocol
Contrast medium dose injection must be adapted to the baby’s weight.
- At our institution,
we use 2 cc per kg (for newborns) of contrast material at 320 mg/ml of concentration.
- We used to inject the contrast material at a rate varying from 1.0 ml/sec to 1.2 ml/sec with a 10 ml saline flush.
- The scan delay was determined using bolus tracking in the descending aorta with a 2 seconds delay after trigger,
repetitive monitoring scans were performed within the lumen of the descending aorta as the region of interest with the threshold of starting the diagnostic scans as 80 HU.
- Mechanical injection of contrast material is the preferred method,
which can provide homogeneous contrast enhancement within the vessels.
- Peripheric venous access is achieved in the pediatric unit or in our department.
Right-arm injection is preferable (but not mandatory),
it doesn’t change the scan injection protocol.
Catheter permeability is checked before the injection.
Scan range
The scan range should be decided according to the patient’s provisional diagnosis.
If the main diagnostic problem is known before MDCT,
it is possible to restrict the scan range to the target area.
However,
because ECHO may occasionally be wrong we still suggest that a neonatal congenital heart disease survey protocol includes the lung apex to the lower margin of the liver.
This scan range can cover all the usual limitations of ECHO such as the aortic arch,
patent ductus arteriosus,
and unusual supracardiac or infracardiac pulmonary venous returns.
Imaging findings
Case 1
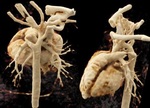
Neonate with vascular ring,non ECG gated CT scan was performed (figure 1 and 2).

Fig. 1: Figure 1. VRT images of complete vascular ring formed by right aortic arch, aberrant left subclavian artery with Kommerell`s diverticulum and left ductus arteriosus. DLP was 5 mGy x cm.
Figure 1.
VRT images of complete vascular ring formed by right aortic arch,
aberrant left subclavian artery with Kommerell`s diverticulum and left ductus arteriosus.
DLP was 5 mGy x cm.

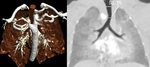
Fig. 2: Figure 2. The same patients, VRT and MinIP images showed compression of the trachea by vascular ring.
Figure 2.
The same patients,
VRT and MinIP images showed compression of the trachea by vascular ring.
Case 2
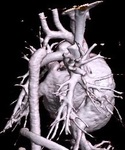
An 15 days old neonate with aortic coarctation (figure 3)

Fig. 3: Figure 3. Volume rendering technique (VRT) image showing aortic coarctation in the isthmus.
Figure 3.
Volume rendering technique (VRT) image showing aortic coarctation in the isthmus.
Case 3
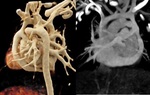
A 4 days old child had total anomalous pulmonary venous connection (figure 4).

Fig. 4: Figure 4. VRT and MIP images and GIF VRT image showed supracardiac total anomalous pulmonary venous connection (TAPVC) draining into persistent left superior vena cava which drains into coronary sinus.
Figure 4.
VRT and MIP images and GIF VRT image showed supracardiac total anomalous pulmonary venous connection (TAPVC) draining into persistent left superior vena cava which drains into coronary sinus.
Case 4
A 17 days old infant had aortopulmonary window (figure 5).

Fig. 5: Figure 5. VRT and transversal images ofa defect between the great vessels(APW).
Figure 5.
VRT and transversal images of a defect between the great vessels (APW).
Case 5
A 2 days old boy with interrupted aortic arch (figure 6).

Fig. 6: Figure 6. VRT images showed interrupted aortic arch between left innominate carotid and left subclavian arteries (type B) with examination protocol.
Figure 6.
VRT images showed interrupted aortic arch between left innominate carotid and left subclavian arteries (type B) with examination protocol.
Case 6
A 11 days old neonate with pulmonary atresia (figure 7).

Fig. 7: Figure 7. VRT images showed pulmonary artery atresia type III with major aortopulmonary collateral arteries(MAPCAs).
Figure 7.
VRT images showed pulmonary artery atresia type III with major aortopulmonary collateral arteries (MAPCAs).