Aims and objectives
Pancreatic adenocarcinoma (PDA) is the fourth cause of cancer-related death,
having a five-years survival rate of 8% [1].
The therapeutic approach to advanced unresectable PDA is systemic cytotoxic chemotherapy.
FOLFIRINOX regimen is currently used as first-line systemic treatment in advanced PDA,
showing a significant improvement in terms of survival compared to gemcitabine in metastatic pancreatic cancer (MPC),
whereas similar results have not yet been proven in locally advanced pancreatic cancer (LAPC) [2].
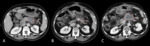
Contrast-enhanced CT (CECT) is the conventional imaging modality for response assessment in this...
Methods and materials
Institutional review board approval and waiver for informed consent was obtained for this retrospective study.
Patients
A computer search in our hospital database was performed,
identifying patients with unresectable PDA treated with FOLFIRINOX regimen between November 2010 and February 2017.
Inclusion criteria were as follows: (1) histological evidence of PDA; (2) unresectability of the pancreatic primary tumour established by a gastro-intestinal multidisciplinary team; (3) FOLFIRINOX therapy duration of at least 1 month or 2 cycles; (4) baseline CECT study including a portal-venous scan of thorax-abdomen-pelvis...
Results
A total of 32 LAPC patients and 35 MPC patients were enrolled in this study.
According to RECIST 1.1 criteria,
in the LAPC group,
responders were 12/32 (37.5%) and non-responders were 20/32 (62.5%) and in the MPC group,
responders were 12/35 (34.3%) and non-responders were 23/32 (65.7%).
In both LAPC and MPC,
CTTA metrics registered at BL and FA showed no significant differences between responders and non-responders.
In the LAPC group,
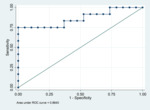
a significant difference (p < 0.001) between responders and non-responders was found for percentage...
Conclusion
Radiological follow-up of cytotoxic systemic treatment for PDA is currently sustained by dimensional criteria that don’t take into account intra-lesional structural changes treatment-related that could reflect drug efficacy.
The aim of this study was to explore the potential relationship between CTTA metrics and their changes throughout therapy follow-up with response categorization by RECIST 1.1.
According to our preliminary results,
CTTA metrics values measured at BL and FA showed no significant difference between responders and non-responders by RECIST 1.1 in both LAPC and MPC groups and...
Personal information
Dr.
Edoardo Raimondi
Radiology Registrar - University of Ferrara - Italy
Clinical Research Fellow - The Royal Marsden Hospital - London - UK
[email protected]
References
Siegel RL,
Miller KD,
Jemal A (2016) Cancer Statistics,
2016,
CA CANCER J CLIN 2016;66:7–30
Conroy T,
Desseigne F,
Ychou M et al,
(2011) FOLFIRINOX versus Gemcitabine for Metastatic Pancreatic Cancer,
N Engl J Med 2011;364:1817-25.
Eisenhauer EA,
Therasse P,
Bogaerts J et al,
(2009) New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1),
European Journal of Cancer 45 (2009) 228–247
Chatterjee D,
Katz MH,
Rashid A et al,
(2011),
Histologic Grading the Extent of Residual Carcinoma Following Neoadjuvant Chemoradiation in Pancreatic...