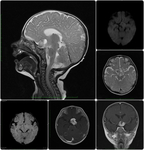
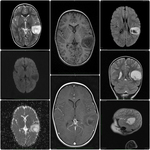
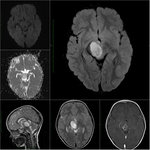
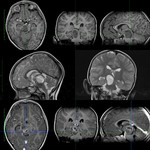
Eight children (mean age 2.1 years,
range 0.2-4.1 years) underwent multiparametric preoperative contrast enhanced MRI in our department between November 2012 and August 2015.
All MRI examinations were performed with a 1.5T MR imaging system (Siemens Magnetom Essenza 1.5T; Siemens Healthcare,
Germany) with a standard head coil.
Intravenous contrast enhancement was made in every case.
In six of eight cases diagnoses of astrocytoma were histologicaly confirmed (two of them were recognized inoperable).
All studies were performed under general anesthesia.
The average total time for research - 28 minutes.
Preoperative Imaging.
On conventional MR imaging,
astocytomas usually show well-marked contrast enhancement,
peritumoral edema,
mass effect,
heterogeneity,
central necrosis,
and sometimes intratumoral hemorrhage.
ADC.
It is not always easy to differentiate low-grade astrocytomas from high-grade ones on the basis of conventional MR imaging findings,
because they can have similar MR features. Measurement of the minimum apparent diffusion coefficient (ADC) can provide desired diagnostic information for preoperative grading of astrocytomas. Low ADC values correlate with poor survival and suggests high-grade type of tumor.
ADC values correlate with grade:
- WHO IV (GBM) = 745 ± 135 x 10-6 mm2/s
- WHO III (anaplastic) = 1067 ± 276 x 10-6 mm2/s
- WHO II (low grade) = 1273 ± 293 x 10-6 mm2/s
- ADC threshold value of 1185 x 10-6 mm2/s sensitivity (97.6%) and specificity (53.1%) in the discrimination of high-grade (WHO grade III & IV) and low-grade (WHO grade II) gliomas.
Contrast enhancement patterns.
On contrast enhanced MRI most of intramedullary astrocytomas show at least some accumulation of contrast material regardless of cell type or tumor grade,
but high-grade type tumors enhancing much more intense.
The types of contrast enhancement patterns of astrocytomas:
1) no enhancement,
2) focal nodular enhancement,
3) patchy enhancement,
4) inhomogeneous diffuse enhancement
5) homogeneous diffuse enhancement.
Typical MR - signal characteristics of Low-grade astrocytomas:
- T1
- isointense to hypointense compared to white matter
- usually confined to the white matters and causes expansion of the adjacent cortex
- T2/FLAIR
- mass-like hyperintense lesion
- always follow the white matter distribution and cause expansion of the surrounding cortex
- cortex can also be involved in late cases in comparison to the oligodendroglioma,
which is a cortical based tumour from the start
- the "microcystic changes" along the lines of spread of the infiltrative astrocytoma is a unique behavior for the infiltrative astrocytoma, however,
it is only appreciated in a few numbers of cases
- high T2 signal is not related to cellularity or cellular atypia,
but rather oedema, demyelination and other degenerative change
- DWI
- no restricted diffusion
- increased diffusibility is the key to differentiate the diffuse astrocytoma from the acute ischemia
- T1 C+ (Gd)
- no enhancement is often the rule but small ill-defined areas of enhancement are not rare; however, when enhancement is seen it should be considered as a warning sign for progression to a higher grade
Typical MR - signal characteristics of High-grade astrocytomas:
- T1
- hypo to isointense mass within white matter
- central heterogenous signal (due to necrosis,
intratumoural hemorrhage)
- T1 C+ (Gd)
- enhancement may be variable but is almost always present
- typically peripheral and irregular with nodular components
- usually completely surrounds necrosis
- T2/FLAIR
- hyperintense lesion surrounded by vasogenic oedema
- flow voids occasionally seen
- GE/SWI
- susceptibility artifact on T2* from blood products (or occasionally calcification)
- low intensity rim from blood product
- incomplete and irregular in 85% when present
- mostly located inside the peripheral enhancing component
- absent "dual rim sign"
- DWI/ADC
- solid component
- elevated signal on DWI is common in solid / enhancing component
- diffusion restriction is typically intermediate similar to normal white matter,
but significantly elevated compared to surrounding vasogenic oedema (which has facilitated diffusion)
- non-enhancing necrotic / cystic component
- the vast majority (>90%) have facilitated diffusion (ADC values > 1000 x 10-6 mm2/s)
- care must be taken in interpreting cavities with blood product
Follow-up imaging.
- Post-operative MRI must be performed within 72 hours after surgery in order to delineate residual tumor from the postsurgical inflammatory changes that are visualized on MRI at this time.
- MRI with contrast of the head should be performed every 3 months during the first year after surgery 1-1.5 and 4-6 weeks after completion of radiation therapy.
- It is advisable to perform an MRI of the spine each year in patients with tumors of high degree,
if at the time of diagnosis was no evidence of a leptomeningeal dissemination; in case of a leptomeningeal dissemination was known in advance,
the frequency of inspections is increased to 3-6 times per year (depending on the chemotherapy protocol)