MDCT Technique for small airway diseases:
Multidetector computed tomography (MDCT) scanners allow diagnosis and monitoring of lung disease at substantially lower radiation doses than with prior scanners. There are two types of CT scanning protocols currently used to assess lung disease:
(1) High-resolution CT (HRCT) imaging, in which thin 0.5–1.5-mm slices are obtained every 0.5, 1, or 2 cm from apex to base for inspiratory scans, and limited, spaced HRCT slices obtained for expiratory scans.
(2) Complete spiral CT imaging covering the entire lung for inspiratory and expiratory scanning. These scanning protocols provide CT datasets to quantify airway and air-trapping measurements.
HRCT in Small airway diseases:
High-resolution CT techniques sample the lung by acquiring thin, 0.5- to 1.5-mm slices every 0.5, 1, or 2 cm with gaps between slices. Scans are usually obtained from the apex to the base of the lungs during inspiration. Because the CT scanner must move and stop the patient for each slice, HRCT requires more time than spiral CT. HRCT scans typically require 2 seconds for each slice, or greater than 40 seconds for lung sizes greater than 20 cm in length. For expiratory scans, a smaller number of HRCT images are obtained, which are either evenly spaced from apex to the base or are obtained at anatomically determined locations. All the CT scan images in this poster are from HRCT chest performed on a 128 slice Toshiba MDCT scanner (CT techniques:128 slicer CT scanner, Toshiba Aquilion, Tokyo, 120kvp / 300mA, 0.5mm thickness) of patients with chronic cough, shortness of breath or clinical suspicion of airway disease.
Imaging patterns of small airway diseases:
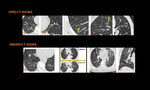
Small airway disease can manifest with direct or indirect signs or both on high resolution computed tomography5. On HRCT images, the considerable thickening of the small airway walls by inflammatory infiltrate and/or luminal and surrounding exudate render the affected small airways directly visible13. When the airways are obliterated by submucosal or peribronchial fibrosis, nodular, linear, or branching peripheral opacities may be seen 14. ( Table 1 ). The imaging detail of each finding of small airway disease is below:
- Bronchiolar wall thickening:
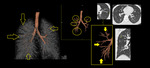
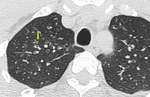
Small airways are not visible on HRCT scan in normal subjects. However, considerable thickening of the bronchiolar walls by inflammatory infiltrate and/or luminal and surrounding exudates render them directly visible 13. Bronchiolar airways should be considered thick walled if the wall is at least twice as thick as that of the normal airway or the internal diameter of the lumen is <80% of its external diameter (Figures 1 & 2).
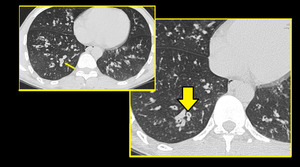
Fig. 2: HRCT images showing Thick walled bronchioles seen in right lower lobe, some with occluded lumen (arrow).
- Intraluminal Plugs:
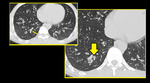
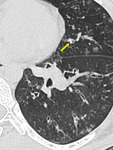
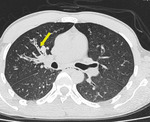
Small variable density intraluminal foci are seen in the bronchioles, which could be mucus impactation, exudates or aspiration (Figures. 3 & 4).
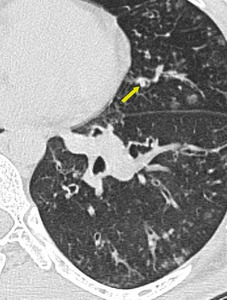
Fig. 3: Patient with infective bronchiolitis showing multiple centrilobular nodules and plugs in lingular bronchiole (arrow).
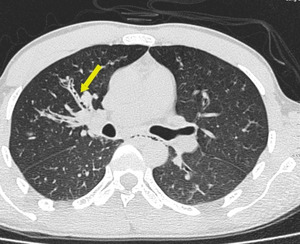
Fig. 4: Mucus impacted bronchioles seen in right upper lobe
- Centrilobular Nodules:
Centrilobular nodules can be classified in two varieties:
a. Poorly defined Centrilobular nodules of ground glass opacity (Figure). They tend to be fairly homogeneous in size. They are inflammatory cellular or fibrosis surrounding the centrilobular bronchiole. In these cases, impactation of bronchiole is typically absent.
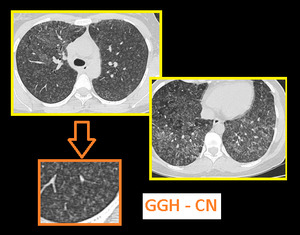
Fig. 5: Soft ground glass haze centrilobular nodules and patchy mosaic attanuation are seen diffusely scattered in both lungs in a patient with hypersensitivity pneumonitis.
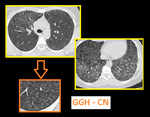
b. Well defined, V or Y shaped denser centrilobular nodules (CN). These are seen due to intraluminal impaction.
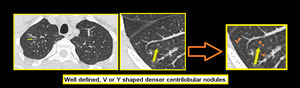
Fig. 6: Centrilobular nodules seen in center of secondary pulmonary lobule.
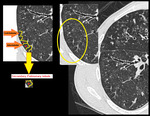
Differential diagnosis of CN nodules include hypersensitivity pneumonitis, respiratory bronchiolitis, RB with ILD (Figure), follicular bronchiolitis, pneumoconiosis, collagen vascular diseases, atypical infections.
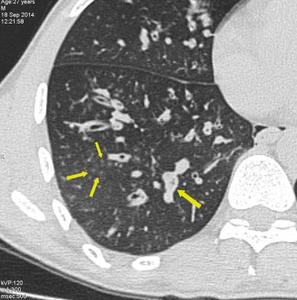
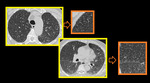
Fig. 8: Small airway disease with multiple centrilobular bronchioles. Magnified image of HRCT showing infective bronchiolitis in a young male. Thick walled bronchioles with mucus plugs and centrilobular nodules are well appreciated.
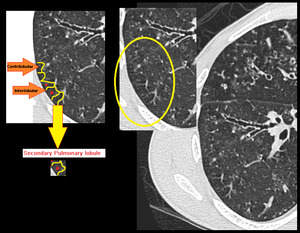
Fig. 7: Centrilobular nodules seen in center of secondary pulmonary lobule.
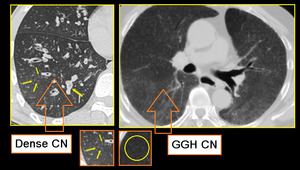
Fig. 9: Small airway disease with multiple centrilobular bronchioles.
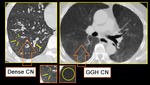
DENSE CN - Magnified image of HRCT showing infective bronchiolitis in a young male. Thick walled bronchioles with mucus plugs and centrilobular nodules are well appreciated (arrows).
GGH CN - Axial section of lungs in a patient with allergic alveolitis. Subtle mosaic low attenuation also noted in background lung suggesting constrictive bronchiolitis.
Important to remember that centrilobular nodules of hypersensitivity pneumonitis (right image) differ from those of infectious cellular bronchiolitis (left image) in that in HP, these are usually diffuse, poorly defined, and of ground-glass attenuation rather than soft-tissue attenuation.
Fig. 10: Axial high resolution CT image of a chronic smoker with respiratory bronchiolitis-interstitial lung disease (RB-ILD). There are bilateral soft ground glass haze like centrilobular nodules. Subpleural interstitial septal thickening is seen bilaterally.
- Tree in Bud Nodules:
This pattern resembles a budding tree where there is dilatation and impactation of centrilobular bronchioles by pus or mucus (the branches) plus associated with peri-bronchiolar inflammation or fibrosis (the buds). Lobular bronchioles are usually not directly visible on CT because their walls are too thin. But they can become visible at the center of the secondary lobule when there is increased soft tissue in or around the bronchioles. Tree-in-bud opacities are generally taken to mean that infection is present but can be seen with any small airway disease (Figure. 10).
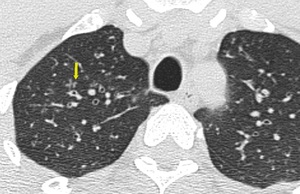
Fig. 11: Tree in bud like branching centrilobular nodules are seen in right upper lobe in a patient with ongoing endobronchial spread of tuberculosis.
MIP-ing is helpful in diagnosing tree in bud nodules. The use of MIP has been proved to increase the number of visualized bronchiolar centrilobular opacities compared with single thin-section CT scan in patients with infectious or inflammatory bronchiolitis (Figure.11).
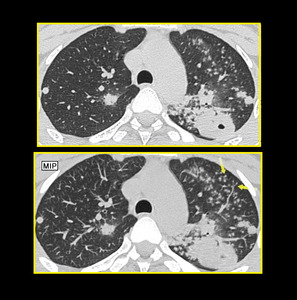
Fig. 12: Axial image at level of aortic arch shows better visualization of branching nodules on MIP image as compared to thin section upper image in a full blown case of tuberculosis. The use of MIP has been proved to increase the number of visualized bronchiolar centrilobular opacities compared with single thin-section CT scan in patients with infectious or inflammatory bronchiolitis.
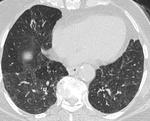
- Bronchiolectasis:
Bronchiolectasis term defines dilated bronchioles visualized in the peripheral 1 to 2 cm of the lung, which is not normal (Figure.12). Bronchiolectasis may be air filled or filled with secretions. On CT images, there might be luminal impactation with bronchiolar wall thickening (Tree in bud). This finding is commonly associated with large airway abnormalities.
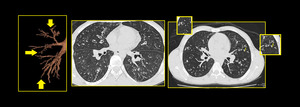
Fig. 13: Bronchiolectasis: Dilated small airways seen in both lungs (arrow).
Few dilated bronchioles in left lower lobe also show wall thickening and are filled with secretions.
- Mosaic attenuation pattern:
Persistent aeration caused by collateral pathways or hyper-aeration from trapped air can produce mosaic attenuation pattern. It is characterized by non-homogenous lung density, i.e. areas that remain relatively lucent interspersed with areas of normal higher lung density (Figure.13).
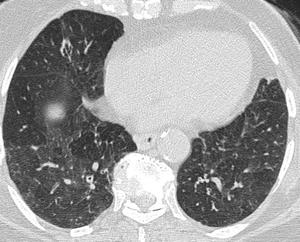
Fig. 14: Bilateral patchy mosaic attenuation in a case of hypersensitivity pneumonitis. Low attenuation areas represent air trapping with attenuated vessels.
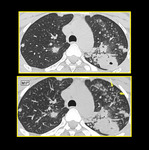
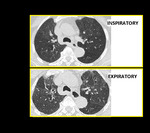
The air trapping and mosaic attenuation pattern is more pronounced, on scans obtained at end-exhalation instead of the more conventional end-inspiration technique 20. Air trapping is indirect sign of obstructive small airways disease that is accentuated on expiratory CT. This means that the low attenuation areas seen on inspiratory scans either persist, increase in number or increase in volume.
When mosaic attenuation/air trapping is the only or predominant finding the differential diagnosis is quite limited i.e. asthma, HP, constrictive bronchiolitis and vasculitis. So, expiratory CT is the key (Figure.14).
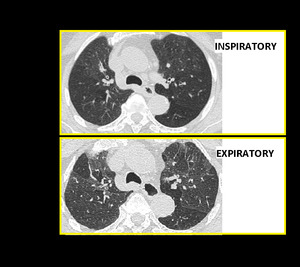
Fig. 15: Air Trapping. Inspiratory and end expiratory axial HRCT images showing patchy mosaic attenuation and accentuation of air trapping on expiratory scan.
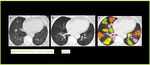
MinIP increases the contrast between areas of normal lung attenuation and areas of lung hypoattenuation facilitating the depiction of mosaic pattern and improve the detection of air trapping (Figure.15).
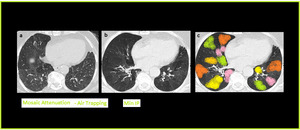
Fig. 16: MinIP in Air Trapping. Axial CT image with Min-IP shows increase in the contrast between areas of normal lung attenuation and areas of lung hypoattenuation as compared to the volume image, facilitating the depiction of mosaic pattern and improves the detection of air trapping. Colored areas of air trapping better visualized with MinIp.
- Chronic small airway disease:
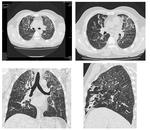
Chronic small airway disease findings on HRCT include vascular attenuation, increased lung volume, subsegmental atelectasis and in late stages even fibrosis (Figure.16,17).
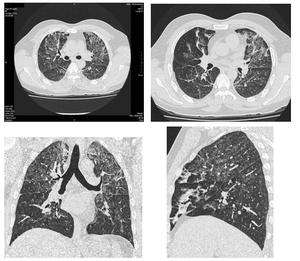
Fig. 17: Multiple axial and reformatted images showing chronic changes of small airway disease. There are bilateral soft ground glass haze nodules, patchy mosaic low attenuation areas with small sized vessels and subsegmental atelectasis.
The pattern and distribution may sometimes give an indication of the underlying etiology (Figure.17).
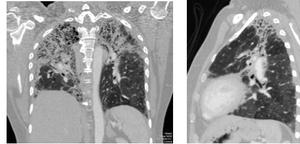
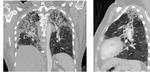
Fig. 18: Thin slice MDCT reformatted images showing interstitial pulmonary fibrosis with interstitial septal thickening, honeycombing and tractional bronchiectasis with upper lung predominance in a patient with chronic hypersensitivity pneumonitis.
The imaging findings are summed up in following table:
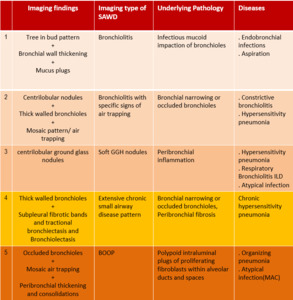
Table 1: Characteristic imaging patterns of small airway diseases on HRCT scan.
Direct and Indirect signs of small airway disease on imaging:
- Direct signs of small airway disease include
- Bronchiolar wall thickening
- Plugs
- Centrilobular Nodules (CN), which are either ill-defined ground glass opacity or well-defined centrilobular branching V or Y nodules
- Tree-in-bud opacities
- Bronchiolectasis
- Indirect signs of small airway disease include
- Mosaic pattern of attenuation (on inspiratory CT scan) and air trapping (on expiratory CT scan and best seen using MinIP)
- Vascular attenuation
- Increased lung volume
- Subsegmental atelectasis
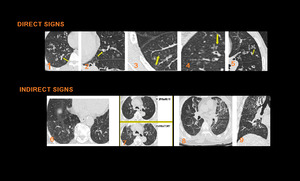
Fig. 19: Direct signs of small airway disease include Bronchiolar wall thickening (1), plugs (2), Centrilobular Nodules (3), which are either ill-defined ground glass opacity or well-defined centrilobular branching V or Y nodules, Tree-in-bud opacities (4),Bronchiolectasis (5). Indirect signs of small airway disease include Mosaic pattern of attenuation on inspiratory CT scan (6) and air trapping on expiratory CT scan (7), Vascular attenuation (8), Increased lung volume & Subsegmental atelectasis (8,9)
Classification of small airway diseases:
A classification of bronchiolitis has been proposed based on etiology and histopathological appearance ( Table)7. However, this classification was largely based on the results of histopathological examination, which requires bronchoscopic or surgical biopsy that cannot be performed in all patients. Devakonda and fellows8 have proposed a diagnostic algorithm based on clinical context and CT findings (
Table)7. However, this classification was largely based on the results of histopathological examination, which requires bronchoscopic or surgical biopsy that cannot be performed in all patients. Devakonda and fellows8 have proposed a diagnostic algorithm based on clinical context and CT findings ( link).This proposal appears useful, considering that most patients with suspected small airways disease do not have surgical lung biopsy, but correlation with pulmonary function tests and other lab findings still required.
link).This proposal appears useful, considering that most patients with suspected small airways disease do not have surgical lung biopsy, but correlation with pulmonary function tests and other lab findings still required.
Proposal for New approach for Classification:
By combining the pathological classification with the radiological findings, we have tried to come up with an easy approach depending on HRCT finding pattern, that could lead to underlying cause of small airway involvement. In our departmental data, four basic radiologic patterns of small airway disease were observed on CT scan ( Table 1 ):
1. Bronchiolitis
2. Bronchiolitis with specific signs of airtrapping
3. Centrilobular ground glass nodules
4. Extensive chronic small airway disease pattern
We divide small airway disease patterns into three main categories and have tried to make it simple (Table.2). So, how to recognize bronchiolitis on HRCT?
- Cellular bronchiolitis is inflammation of the bronchiolar wall with intraluminal exudates. HRCT images would show bronchiolar wall thickening, plugs, Tree in bud pattern and centrilobular nodules.
- Constrictive bronchiolitis is peri-bronchiolar fibrosis with extrinsic compression and obliteration of the airway. HRCT scan would show bronchiolar wall thickening, bronchial narrowing/occlusion and mosaic air trapping.
- Bronchiolitis obliterans with intraluminal polyps or BOOP. There are polypoid intraluminal plugs of proliferating fibroblasts in alveolar ducts & spaces thus CT images would show acinar ground glass haze, consolidations with bronchiolectasis and plugs.
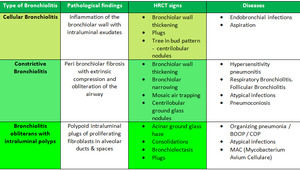
Table 2: showing combined classification of pathological types and radiological findings. We combined the radiological and pathological classification and propose a simple approach.
Limitations and Differentials:
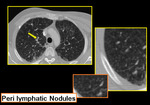
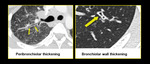
Few limitations prevail like not all patients could hold breath for expiratory scans. While reporting HRCT for small airway diseases certain false positives should also be considered like differentiating peri-bronchial thickening from bronchiolar wall thickening especially in advanced peri-lymphatic disease (Figure.19, 20).
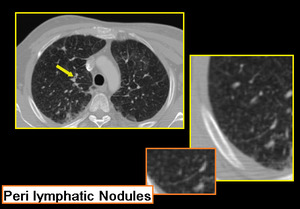
Fig. 20: Sarcoidosis with infiltrative ground glass haze. Bilateral patchy areas of mosaic attenuation. Few thick walled bronchioles seen in right apical segment. Nodular interstitial thickening in posterior segment of right upper lobe.
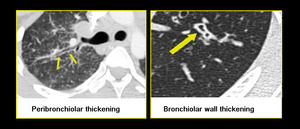
Fig. 21: Peri-bronchial thickening of sarcoidosis versus bronchiolar wall thickening in infective bronchiolitis.
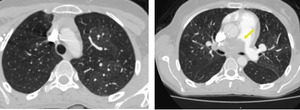
Fig. 22: Patchy mosaic areas in both lungs with attenuated vessels in a case of chronic pulmonary embolism. Thrombus is appreciated in right main pulmonary artery (arrow).
Over-reporting of normal expiratory scans, which show few small subpleural air trapped areas, should be avoided.
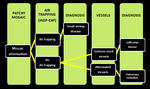
Mosaic pattern - Challenge to Radiologist:
Mosaic pattern of lung attenuation presents a challenge to radiologist when deciding which the abnormal areas of the lung are; those of low attenuation, those of high attenuation, or both. We followed a simple algorithm considering three categories of diseases, known to cause a CT mosaic pattern of lung attenuation i.e. Small-airway disease, Vascular lung disease, Infiltrative disease (Figure.19,20,22).
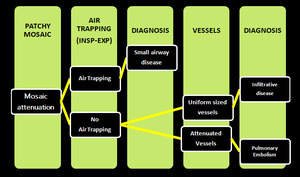
Fig. 23: showing different possible diseases with mosaic attenuation pattern on HRCT scans.